Board of Patent Appeals and Interferences
Patent and Trademark Office (P.T.O.)
*1 EX PARTE HANS BLATTNER AND ANGELO STORNI
Appeal No. 639-05
May 14, 1987
Heard: April 30, 1987
Application for Patent filed September 29, 1980, Serial No. 191,728, a continuation of Serial No. 961,324, filed November 16, 1978, abandoned; which is a continuation-in-part of Serial No. 798,204, filed May 18, 1977, abandoned. Azatetracyclic Compounds.
Irving N. Feit, et al., for Appellants
Primary Examiner--Richard A. Schwartz
Before Winters, Sofocleous, and Kimlin
Winters
Examiner-in-Chief
This appeal arises from the examiner's final rejection of claims 6 through 20, which are all of the claims in the application. In the examiner's answer, page 2, the examiner indicates that the subject matter defined in claims 8 and 10 is allowable over the prior art of record. This leaves for our review claims 6, 7, 9 and 11 through 20.
Claims 17, 11 and 14 are representative.
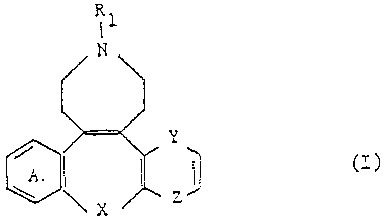
17. An azatetracyclic compound of the formula
wherein
R1 represents hydrogen, lower alkyl, cycloalkyl-lower alkyl of not more than 10 carbon atoms, lower alkenyl, lower alkynyl, (di-lower alkylamino)-lower alkyl, hydroxy lower alkyl, alkoxy lower alkyl, alkanoyloxy lower alkyl having 4 to 11 carbon atoms, lower alkylthio-lower alkyl, phenyl-lower alkyl, phenyl- lower alkyl substituted by halogen with an atomic number up to 35, lower alkyl, lower alkoxy, methylenedioxy, and trifluoromethyl, or lower alkanoyl, the ring A is unsubstituted or substituted by halogen with an atomic number up to 35, lower alkyl, hydroxyl, lower alkoxy, alkanoyloxy having 1-3 carbon atoms, lower alkylthio, trifluoromethyl or cyano, X represents 0, S, methylene, a direct bond or a divalent radical of the partial formula

in which R3 represents hydrogen or lower alkyl, and one of the radicals Y and Z represents vinylene or S and the other represents a direct bond, and the acid addition salts thereof.
11. A pharmaceutical composition useful in the treatment of states of agitation in a warmblooded animal comprising a therapeutically effective amount of an azatetracyclic compound according to claim 17, or a pharmaceutically acceptable acid addition salt thereof together with a pharmaceutical carrier.
14. A method of treating states of agitation in a warmblooded animal which comprises administering to said animal a therapeutically effective amount of a compound according to claim 17, or a pharmaceutically acceptable acid addition salt thereof.
The single prior art reference relied on by the examiner is:
van der Burg 4,002,632 Jan. 11, 1977
The sole issue presented for review is whether the examiner correctly rejected claims 6, 7, 9 and 11 through 20 under 35 U.S.C. 103 as unpatentable over van der Burg. More specifically, the issue is whether the examiner has established that the claimed subject matter would have been prima facie obvious in view of the disclosure of van der Burg.
OPINION
*2 We shall not sustain this rejection.
In obviousness rejections based on close similarity in chemical structure, the prima facie case of obviousness rises from the expectation that compounds similar in structure will have similar properties. In re Payne, 606 F.2d 303, 203 USPQ 245, 254 (CCPA 1979); In re Gyurik, 596 F.2d 1012, 201 USPQ 552, 557 (CCPA 1979); In re Hoch, 57 CCPA 1292, 428 F.2d 1341, 166 USPQ 406, 409 (1970). The examiner here takes the position that appellants' claimed compounds, pharmaceutical compositions containing same, and method of treating states of agitation by administering same would have been obvious in view of the disclosure of van der Burg. Specifically, the examiner asserts that appellants' azepine compounds which contain a 7-membered ring would have been prima facie obvious in view of the closely related pyrrolidino and piperidino compounds disclosed by the prior art reference. Pyrrolidino and piperidino compounds contain a 5- and 6-membered ring, respectively. We disagree with the examiner's position.
Van der Burg discloses that the known pyrrolidino and piperidino compounds possess diametrically opposite utilities. See the van der Burg patent, column 1, lines 43 through 62. As described therein, the pyrrolidino derivatives are useful in treating conditions of stress and agitation whereas the piperidino compounds are useful in treating depressed patients. This fact is not in dispute. As acknowledged by the examiner in his answer, page 4, a person having ordinary skill in the art 'knows that the 5- and 6-membered compounds also possess opposite utilities.' Where, as here, the pyrrolidino and piperidino 'ring homologs' disclosed by the prior art possess diametrically opposite utilities, this salient fact substantially undermines the examiner's asserted prima facie case of obviousness, which 'rises from the expectation that compounds similar in structure will have similar properties.' In re Gyurik, supra, 201 USPQ at 557. Quite clearly, the very reference relied on by the examiner teaches that compounds similar in structure in this art do not possess similar properties.
We agree with appellants that a person having ordinary skill in the art, given only the van der Burg patent but not appellants' disclosure, would not have had sufficient basis to predict what, if any, utility appellants' azepine compounds might possess. What utility would a person having ordinary skill in the art have expected for the azepine or 7-membered ring compound? A transquilizer? An anti-depressant? A compound which is neither because the depressant and anti-depressant effects counteract each other?
In conclusion the examiner's prima facie case of obviousness is based on the expectation that compounds which are similar in structure will have similar properties. The very prior art reference here relied on by the examiner, however, undermines the prima facie case because the reference teaches that compounds similar in structure in this art do not possess similar properties. Accordingly, the examiner's decision is reversed.
REVERSED
BOARD OF PATENT APPEALS AND INTERFERENCES
Examiner-in-Chief
Michael Sofocleous
Examiner-in-Chief
Edward C. Kimlin
Examiner-in-Chief
<< Return to Board of Patent Appeals and Interferences Index