Board of Patent Appeals and Interferences
Patent and Trademark Office (P.T.O.)
*1 QUINOLINECARBOXYLIC ACID DERIVATIVES
Patent Interference Nos. 101,789; 101,790; 101,839
August 29, 1990
Final Decision Heard: June 21, 1990
Applications of Tsutomu Irikura, Seigo Suzue, Satoshi Murayama, Keiji Hirai and Takayoshi Ishizaki, filed May 20, 1985, Serial No. 735,987.
Applications of Tsutomu Irikura, Seigo Suzue, Keiji Hirai and Takayoshi Ishizaki, filed July 25, 1985, Serial No. 758,727.
Applications of Tsutomu Irikura, Seigo Suzue, Keiji Hirai and Takayoshi Ishizaki, filed November 4, 1985, Serial No. 794,378.
Application of Uwe Petersen, Klaus Grohe, Hans-Joachim Zeiler and Karl G. Metzger, filed May 17, 1985, Serial No. 735,500.
Norman F. Oblon, Stanley P. Fisher, Marvin J. Spivak, C. Irvin McClelland, Gregory J. Maier, Arthur I. Neustadt, Robert C. Miller, Richard D. Kelly, James D. Hamilton, Eckhard H. Kuesters, Robert T. Pous and Charles L. Gholz. Oral Argument by Steven Kelber
Arnold Sprung, Leonard Horn, Nathaniel Kramer, James F. Woods, Joseph G. Kolodny and Ira J. Schaefer. Oral Argument by Leonard Horn.
Before Torchin, Downey and W. Smith
Examiners-in-Chief
Downey
Examiner-in-Chief
Final Decision
Before us are three interferences, Nos. 101,789, 101,790 and 101,839, involving the application of Petersen et al. (SN. 735,500) and three Irikura et al. applications: (1) Irikura, Suzue, Murayama, Hirai and Ishizaki (SN. 735,987 involved in Interference No. 101,789), (2) Irikura, Suzue, Hirai and Ishizaki (SN. 758,727 involved in Interference No. 101,790) and (3) Irikura, Suzue, Hirai and Ishizaki (SN. 794,378 involved in Interference No. 101,839).
Both parties filed briefs and appeared through counsel at final hearing. The party Irikura et al. filed identical briefs and reply briefs; whereas Petersen et al. filed separate, albeit a substantially identical main brief in each interference. For convenience and for consideration of the substantially identical issues involved, we have consolidated the decisions in the three interferences into a single opinion.
The Peterson et al. application is assigned to Bayer Aktiengesellschaft and the three Irikura et al. applications are subject to an unrecorded assignment to Kyorin Pharmaceutical Company, Ltd.
The subject matter at issue in each interference relates to certain quinoline carboxylic acid derivative compounds useful as antibacterial agents.
The count of 101,789 reads as follows:
Count 1
A compound of the formula (I):
wherein R is hydrogen atom or methyl group; the hydrates or the pharmaceutical acceptable acid addition or alkali salts thereof.
The claims of the parties which correspond to the above count are:
Irikura et al: Claims 1 and 3.
Petersen et al.: Claims 1-15.
The count of 101,790 reads as follows:
Count 1
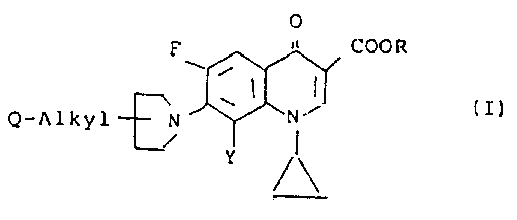
A compound of the formula (I):
wherein R1 is a hydrogen atom or a lower alkyl group, R2 is a hydrogen atom or a methyl group; or the hydrates or pharmaceutically acceptable acid addition or alkali salts thereof.
*2 The claims of the parties which correspond to the above count are:
Irikura et al: Claims 4-9.
Petersen et al: Claims 1-15.
The count of 101,839 reads as follows [FN1]:
Count 2
A compound of the formula (I):
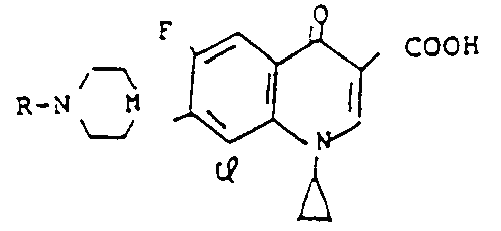
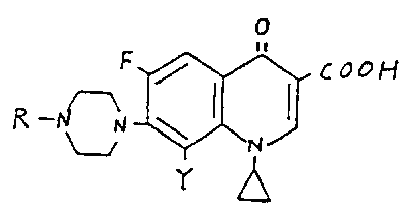
wherein R is a hydrogen atom or a lower alkyl group, Q is an amino-containing radical and Y is a chlorine atom or a bromine atom; the hydrates or the pharmaceutically acceptable acid addition or alkali salts thereof.
The claims of the parties which correspond to the above count [FN2] are:
Irikura et al: Claims 1, 4 and 14-20.
Petersen et al: Claims 1-3 and 12-15.
In response to an order to show cause why judgment pursuant to 37 CFR 1.640(d)(1) and (3) should not be entered against them, Irikura et al. requested a final hearing on the issues presented in the Examiner-in-Chief's (EIC's) Decision on Motions with respect to five particular items therein as follows:
1. Irikura et al. Motion I [designated by the EIC as I.A(1) and I.A(2)] to consolidate the three interferences or alternatively, for judgment that the counts of copending interferences 101,790 and 101,839 are not patentably distinct from count of interference 101,789;
2. Irikura et al. Motion II [designated by the EIC as I.B(1)] to substitute Count A for existing count 1, or if Count A is considered too narrow, to substitute Count B or C for existing count 1;
3. Irikura et al. Motion III [designated by the EIC as I.C] for judgment on the grounds that Petersen et al. claims 1-8 and 12-15 are unpatentable to them under 35 USC 103 over prior art;
4. Irikura et al. Motion VI [designated by the EIC as I.F] to amend the Irikura et al. applications and for benefit of the filing date of earlier filed United States Application Serial No. 677,015;
5. Irikura et al. Motion VII [designated by the EIC as I.G] for benefit of the filing date of Japanese application 232,560/1983.
The EIC dismissed Item 1 and denied Items 2-5. (See Paper Nos. 56, 55, and 56, respectively).
In addition, Irikura et al. requested a testimony period to submit evidence in support of Items 2 and 3, which request was initially denied by the EIC (Paper Nos. 70, 69 and 70); however on reconsideration, the evidence [FN3] submitted with the motions was permitted entry. (Paper Nos. 76, 75 and 76).
Item 1.
EIC's Dismissal of the Irikura et al. Motion I.
Irikura et al. moved to consolidate the three interferences, or in the alternative, for entry of judgment since the counts in the three interferences were patentability indistinct. The EIC dismissed the motion holding that such consolidation is not provided for by the Rules of Practice and that it is not proper for the Board to render an advisory opinion as to the patentability of the counts in the three interferences.
Irikura concedes that the rules of practice, 37 CFR, do not provide for consolidation of the three interferences but they contend that their presentation of a motion for judgment was within the rules pursuant to § 1.633(a) and therefore the EIC improperly dismissed the alternative motion for judgment.
*3 We can review the EIC's interlocutory order only to the extent of determining whether it "was manifestly erroneous or an abuse of discretion"D'. See 37 CFR 1.655(a).
We have reviewed the EIC's order and find it to be proper, without any abuse of discretion or manifest error. The granting of a 37 CFR § 1.633(a) motion for judgment results in the entry of a "judgment"D' to a party, which constitutes a final agency determination that the losing party is not entitled to a patent with claims corresponding to a lost count. Orikasa v Oonishi, 10 USPQ2d 1996 n. 20 (Comm'r 1989). The Irikura et al. request that the EIC determine whether the individual counts were patentably distinct in the three interferences would not, and could not, result in the entry of a judgment to Irikura et al. in their favor.
Accordingly, the Irikura et al. request that we overrule the EIC is denied.
Item 3.
Irikura et al. Motion III for Judgment That Petersen et al. Claims 1-8 and 12- 15 Are Unpatentable Under 35 USC 103 over Prior Art [FN4].
We agree with Irikura et al. that the examiner erroneously determined that the Zeiler declaration (IR 12-21) [FN5] was sufficient to overcome the prima facie case of obviousness over Grohe (I) (Grohe) (IR 31-43). [FN6] Accordingly, we hold that the EIC erred in determining that claims 1-8 and 12-15 in Interference Nos. 101,789 and 101,790 and claims 1-3 and 12-15 in Interference No. 101,839 were allowable over Grohe for the following reasons.
Grohe shows quinoline carboxylic acid derivatives that differ from the Petersen et al. claimed quinoline carboxylic acid derivatives in having a 6,8- difluoro substituent whereas the Petersen et al. claims embrace quinoline derivatives having 6,8-dichloro, 6-fluoro-8-chloro, and 6-chloro-8-fluoro substituents. See Examples 1-5 and 10-12 (IR 38-39).
There is no dispute that Grohe establishes a prima facie case of obviousness with respect to the Petersen et al. claims and that the showing of Zeiler in Table 2 (IR 15) shows that the 6-fluoro-8-chloro quinoline carboxylic acid derivative of the count provides an unexpected advantage over the 6,8-difluoro quinoline carboxylic acid derivative of Grohe. However, the Zeiler declaration provides no comparison between the 6,8-difluoro quinoline carboxylic acid derivative of Grohe and the 6,8-dichloro and the 6-chloro-8-fluoro quinoline carboxylic acid derivatives claimed by Petersen et al.. It is well settled that once a prima facie case of obviousness is established, the burden of going forward with proofs of patentability shifts to the applicant. In re Rinehart, 531 F.2d 1048, at 1051, 189 USPQ 143, 147 (CCPA1976). It is equally well settled that the comparison must be with the closest prior art, In re De Blauwe, 736 F.2d 699, 222 USPQ 191 (Fed.Cir.1984); In re Burckel, 592 F.2d 1175, 201 USPQ 67 (CCPA1979; and that the comparison must be commensurate with the scope of the claims, In re Grasselli, 713 F.2d 731, 218 USPQ 769 (Fed.Cir.1983).
*4 The Petersen et al. claims embrace both unobvious and obvious subject matter because the Zeiler declaration establishes the 6-fluoro-8-chloro quinoline carboxylic acid derivative provides an unexpected advantage but fails to make any showing between the 6,8-dichloro and 6-chloro-8-fluoro quinoline carboxylic acid derivatives and the 6,8-difluoro quinoline carboxylic acid derivatives of Grohe. After being apprised of this position, Petersen et al., under the rules of practice, 37 CFR 1.639 [FN7], were permitted to submit evidence to establish that the claimed 6,8-dichloro and 6-chloro-8-fluoro substituted quinoline carboxylic acid derivatives in their specification provided unexpected results. Petersen et al. did not follow the provisions of § 1.639 by presenting additional evidence with their opposition or by providing a statement of belief that testimony was necessary and a description of the nature of the testimony needed. Accordingly, the Petersen et al. showing is not sufficiently demonstrative regarding nonobviousness to overcome the evidence of obviousness adduced by the examiner since it does not afford a basis upon which to conclude that unexpected results are obtained with respect to the claimed 6,8-dichloro and 6-chloro-8-fluoro substituted quinoline carboxylic acid derivatives. Hence, we agree with Irikura et al. that the EIC should have granted the Irikura et al. motion for judgment against Peterson et al. under § 1.633(a). Whereas claims 1-8 and 12-15 of Interference Nos. 101,789 and 101,790 and claims 1-3 and 12-15 of Interference No. 101,839 are unpatentable under 35 USC 103 over Grohe, judgment shall be entered against Petersen et al. as to these claims.
Item 2.
Irikura et al. Motion II to Substitute Count A, B or C for Count 1
Irikura et al. moved to substitute Count A, B or C for the existing count in each of the three interferences urging that the existing counts in each of the interferences are patentably indistinct and that the count in each interference should have been broader. In their motion, they urged that the Petersen et al. claims in each of the interferences, which corresponded to the count, were identical and that species claims within the scope of one count of one interference were designated as corresponding to the count in 101,790 and 101,839. They further urged that the obviousness-type double patenting rejection in application SN. 735,987 over the claims of copending application 758,727 (IR 105-106) and their acquiescence thereto (IR 107-108) furnishes evidence of patentable indistinctness. At oral hearing, Irikura et al. asserted that terminal disclaimers were filed in each of their involved applications. [FN8]
We have considered each argument of Irikura et al. but are not persuaded that the motion to substitute the proposed counts should have been granted. At the outset of this interference proceeding, the examiner determined that the three Irikura et al. applications defined separate patentable inventions and also that there should be three separate interferences. It is presumed that the examiner is correct Orikasa v. Oonishi, supra; and the burden of persuasion falls upon Irikura et al., who urges the contrary, to prove otherwise.
*5 While it is true that Petersen et al. application claims 9-11 are designated as corresponding to the count in 101,789 and 101,790, such designation does not clearly establish that the proposed counts define the same patentable invention. Moreover, failure by Petersen et al. to move to correct any inconsistency in their designation of claim correspondence does not and will not inure to the benefit of Irikura et al..
Although the examiner made an obviousness-type double patenting rejection in SN 735,987, the Irikura et al. response expressed the view that such rejection was improper (IR 107-108). In any event, the examiner dropped the obviousness-type double patenting rejection prior to initiating the interference which clearly indicates that the examiner finally considered these applications to contain separate patentable inventions. Orikasa v. Oonishi, supra.
Irikura et al. urge that the patents to Grohe and Culbertson would establish that amino substituents in the 7-position are equivalent. However, submission of patents accompanied by attorney argument will not take the place of testimony by experts with regard to equivalency. Meitzner v. Mindick, 549 F.2d 775, 193 USPQ 17 (CCPA1977).
In our view, Irikura et al. has failed to sustain their burden of establishing that the primary examiner erred in setting up three interferences and that the EIC improperly denied their motion.
Items 4 and 5
Irikura et al. Motions VI and VII for Benefit of the Filing Dates of Earlier Filed U.S. Patent and Japanese Applications
The EIC correctly denied these motions.
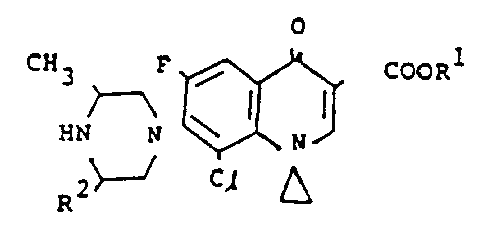
In Motion VI, Irikura et al. moved for benefit of U.S. application SN. 677,015, filed in the names of Irikura, Suzue, Murayama, Hirai and Ishizaki and directed to a quinoline compound having the following formula:
wherein R is hydrogen, methyl, p-nitrobenzyl or p-aminobenzyl and Y is chlorine, fluorine or methyl; hydrates and pharmaceutically acceptable salts thereof.
In Motion VII, Irikura et al. moved for benefit of Japanese application No. 232,560 also filed in the names of Irikura, Suzue, Murayama, Hirai and Ishizaki and directed to a quinoline compound having a generic formula which is the same as that of U.S. SN 677,015 except that R, as defined, does not include a p-nitrobenzyl substituent and a cyclopropyl substituent, which while disclosed in Examples 8-9, is not depicted therein. See page 1 of translation.
In order for Irikura et al. to be entitled to benefit of U.S. Patent Application S.N. 677,015 under 35 USC 120, the previously filed application must comply with the requirements of the first paragraph of 35 USC 112. The statute specifies that the previously filed application provide a written description of the present invention in full, clear, concise and exact terms and it must enable any person skilled in the art to make and use the invention. In re Ahlbrecht, 435 F.2d 908, 168 USPQ 293 (CCPA1971).
*6 In order for Irikura et al. to be entitled to benefit of Japanese Patent Application No. 232,560 under 35 USC 119, the disclosure of the Japanese application must comply with the first paragraph of 35 USC 112 in the same manner as a United States application seeking benefit under § 120. Olson v. Julia, 209 USPQ 159 (Bd.Pat.Int.1979) citing Kawai v. Metlesics, 480 F.2d 880, 178 USPQ 158 (CCPA1973). Additionally, 35 USC 119 requires that the previously filed foreign application must have been filed within twelve months prior to the filing date of the United States application and it must have been filed on behalf of the same inventive entity as in the domestic application. Olson v. Julia, supra.
There is no dispute that the benefit applications comply with the enablement requirement of 35 USC 112. The issue herein is whether the earlier filed applications satisfy the description requirement of the statute. It has been held that the description requirement is satisfied if the benefit application discloses a single species within the count. However, all of the named species in U.S. application SN. 677,015 and Japanese application No. 232,560 are 6, 8- difluoro quinoline carboxylic acid derivatives (see U.S. application examples 8-17 and Japanese application examples 8-9). Hence, there are no species disclosed in either the U.S. or Japanese application which fall within any of the counts in the three interferences.
Additionally, with regard to Interference Nos. 101,790 and 101,839, the counts require a quinoline compound having 3-methylpiperazinyl substituent and a alkylaminopyrrolidinyl substituent in the 7-position, respectively, and neither the asserted U.S. nor Japanese benefit applications provide descriptive support for these substituents. Moreover, the Japanese application, filed in the name of Irikura, Suzue, Murayama, Hirai and Ishizaki, does not have the same inventive entity as the domestic applications involved in 101,790 and 101,839, filed in the name of Irikura, Suzue, Hirai and Ishizaki, Olson v. Julia, supra.
With respect to Interference No. 101,789, the U.S. and Japanese Patent Applications do not constitute an adequate written description for a species within the count. Flynn v. Eardley, 479 F.2d 1393, 178 USPQ 288 (CCPA1973); Fields v. Conover, 443 F.2d 1386, 170 USPQ 276 (CCPA1971); In re Ruschig, 379 F.2d 990, 154 USPQ 118 (CCPA1967); Biel v. Chessin, 347 F.2d 898, 146 USPQ 293 (CCPA1965) and Watson v. Bersworth, 251 F.2d 898, 116 USPQ 79 (CADC1958). Although it is not necessary that the now claimed subject matter be described in ipsis verbis in the benefit application in order to satisfy the description requirement of 35 USC 112, there must nonetheless be some suggestion or guidance by which a person having ordinary skill in the art would arrive at a species within the count. After reviewing the disclosures of the asserted U.S. and Japanese benefit applications, we find no species within the count nor any direction in the applications which constitutes an adequate written description of a compound within the scope of the count of 101,789.
*7 Irikura et al. urge that since the genus disclosed in the U.S. and Japanese benefit applications sets forth a limited number of compounds, it leads the skilled worker to such a limited number of compounds as to effectively describe a species within the count. We cannot agree. Since it has been determined that the 6-fluoro-8-chloro quinoline carboxylic acid derivative is patentably distinct from the 6, 8-difluoro quinoline carboxylic acid derivative, it is necessary that the previously filed benefit application specifically describe the 6-fluoro-8-chloro-quinoline species. Bigham v. Godtfredsen, 857 F.2d 1415, 8 USPQ2d 1266 (Fed.Cir.1988) (disclosure of halogen genus does not provide descriptive support for bromine species when bromine is asserted to be patentably distinct from other halogen species).
We are in accord with the EIC's decision that the U.S. and Japanese benefit applications do not comply with the requirements of 35 USC 112, first paragraph, description requirement. Hence, the motions for benefit of the filing date of U.S patent application, SN. 677,015, and Japanese application, No. 232,560, were properly denied.
Irikura et al. Request for a Testimony Period
Irikura et al. urge that they were denied the opportunity with regard to Motions II and III to enter into the record the evidence relied upon to support the motions. We have reviewed the record and find that Irikura et al. have no basis for this position since the evidence submitted was entered into the record. See the Requests for Reconsideration (Paper Nos. 76, 75 and 76), IR 12-16 and IR 22-108.
Irikura et al. also urge that they were denied a testimony period in which to supplement their original motion III, i.e., they were not permitted to enter into the record their attachments to motion III in affidavit or declaration form and they were not permitted to supplement this motion with expert testimony.
We find no error in the EIC's holding. Since the Irikura et al. motion III has been granted, it would appear that review of the denial of a testimony period would be rendered moot. However, in the interest of completeness the following comments are made.
37 CFR 1.639(a) requires evidence to be filed, or to be on file in the Patent and Trademark Office, to support a preliminary motion under 37 CFR 1.633(c) or an opposition to such a motion. A good faith effort must be made to submit evidence to support a preliminary motion or opposition when the evidence is available. Orikasa v. Oonishi, supra. Note the commentary 49 F.R. 48424, at 48442 (Dec. 12, 1984), 1050 O.G. 393 at 411 (Jan. 19, 1985); corrected, 50 F.R. 23122 (May 31, 1985), 1059 O.G. 27 (Oct. 22, 1985) indicating that if affidavits cannot be timely prepared and filed with a motion, the moving party may wish to take advantage of Rule 1.639(c) which requires a party to specify any testimony needed to resolve the motion. Irikura et al. did not avail themselves of this rule nor the provisions of § 1.645.
Decision in 101,789
*8 Judgment in 101,789 is entered against Petersen et al., the senior party, on the ground that the subject matter of claims 1-8 and 12-15 is not patentable to them under 35 USC 103. Petersen et al. are not entitled to a patent containing their claims 1-8 and 12-15 corresponding to the count.
Judgment in 101,789 as to priority of invention of the subject matter of claims 9-11 corresponding to count 1 is awarded to Petersen et al.. Accordingly, Irikura is not entitled to a patent containing their claims 1 and 3 corresponding to count 1 and Petersen et al. are entitled to a patent containing their claims 9-11.
Decision in 101,790
Judgment in 101,790 is entered against Petersen et al., the senior party, on the ground that the subject matter of their claims 1-8 and 12-15 are not patentable to them under 35 USC 103. Petersen et al. are not entitled to a patent containing their claims 1-8 and 12-15 corresponding to the count.
Judgment in 101,790 as to priority of invention of the subject matter of claims 9-11 corresponding to count 1 is awarded to Petersen et al.. Accordingly, Petersen et al. are entitled to a patent containing their claims 9-11 corresponding to the count and Irikura et al. are not entitled to a patent containing their claims 4-9 corresponding to count 1.
Decision in 101,839
Judgment in 101,839 is entered against Petersen et al. on the ground that the subject matter of their claims 1-3 and 12-15 are not patentable to them. Accordingly, Petersen et al. are not entitled to a patent containing their claims 1-3 and 12-15. Judgment as to priority of invention of the subject matter in the count is entered against Irikura et al. and they are not entitled to a patent containing their claims 1, 4 and 14-20.
BOARD OF PATENT APPEALS AND INTERFERENCES
Norman G. Torchin
Examiner-in-Chief
Mary F. Downey
Examiner-in-Chief
William F. Smith
Examiner-in-Chief
FN1. Interference 101,839 was redeclared on June 12, 1989 in Paper No. 57.
FN2. The Petersen et al motion to substitute the count (Motion B.) was granted. See Paper No. 56.
FN3. Prior art publications U.S. Patents 4,638,067 and 4,556,658 and European Application No. 0126355, the decisions of the Primary Examiner in the involved applications regarding the obviousness-type double patenting rejection, the Zeiler declaration and Exhibit B attached to Motion III.
FN4. At final hearing the Board considers this motion as it applies to the involved claims in each interference. In Interference No. 101,839, only claims 1-3 and 12-15 of Petersen et al. are designated as corresponding to the count.
FN5. Hereinafter, "IR"D' refers to the page(s) of the Irikura et al. Record.
FN6. Motion (III) by Irikura et al. also refers to a rejection premised upon Matsumoto and Grohe (II) in "confirmation with Wise"D' (Paper No. 13). However, the Irikura et al. position with regard to this rejection is not set forth in the brief. Matters not raised in the brief are ordinarily regarded as abandoned. Photo v. Lunkenheimer, 225 USPQ 948 (Bd.Pat.Int.1984).
FN7. 37 CFR § 1.639 states in part:
(a) Proof of any material fact alleged in a motion, opposition, or reply must be filed and served with the motion, opposition, or reply . . . .
(b) Proof may be in the form of patents, printed publications, and affidavits.
(c) When a party believes that testimony is necessary to support or oppose a preliminary motion under § 1.633..., the party shall describe the nature of the testimony needed. If the examiner-in-chief finds that testimony is needed to decide the motion, the examiner-in-chief may grant appropriate interlocutory relief and enter an order authorizing the taking of testimony and deferring a decision on the motion to final hearing.
FN8. A terminal disclaimer was filed in SN. 735,987 with regard to non-involved application, SN. 677,015. However, it was not acceptable because it was signed by an officer of Kyorin Pharmaceutical and not by the owner of record.
Notwithstanding the Irikura et al. assertion, no terminal disclaimers were filed in the involved applications relative to one another.
<< Return to Board of Patent Appeals and Interferences Index